|
Achievements of the Bohr Modelįollowing are the achievements of Bohr’s atomic model. This equation is known as the Bohr quantisation postulate or principle of quantisation of angular momentum. Where n = an integral (called principal quantum number), which denotes the number of the orbit in which the electron is revolving and equals 1, 2, 3, etc. If □ is the velocity and m is the mass of the electron in an authorised orbit of radius r, then, The permitted orbits are those for which the angular momentum of an electron is an integral multiple of h/ 2□, where h = Planck’s constant. The released energy appears as a spectral line in the emission spectrum.Ĩ. This equation is known as Bohr’s frequency condition. Where □ = frequency of the radiation absorbed or released by the electron and As the distance between the energy level and nucleus increases, the energy associated with a certain energy level increases. As the electron in these orbits is associated with a certain amount of energy, these circular paths are also called energy levels or energy shells.ģ. It revolves throughout the nucleus in a fixed concentric circular path called orbits or shells. 
The electron in an atom behaves as a material particle. This nucleus is responsible for almost the entire mass of the atom.Ģ. Due to the existence of protons, the nucleus is positively charged. The neutrons and protons are located in a small nucleus at the centre of the atom. The atom, on the whole, is electrically neutral due to an identical number of negative electrons and positive protons. – Electrons possess a negative charge, neutrons have no charge, whereas protons have a positive charge. An atom comprises three particles, i.e., neutrons, protons, and electrons.In contrast, it will have less energy if situated near the nucleus. Bohr concluded that an electron would have more energy if it is far from the nucleus. However, to account for the stability of the atom, he introduced the concept of the stationary orbit.īohr’s Atomic model explains that a small positively charged nucleus is surrounded by negatively charged electrons revolving in fixed orbits. Experimental results of atomic spectra can explain only by assuming quantised electronic energy levels in atoms.īohr retains the essential features of the Rutherford model of the atom.The dual personality of electromagnetic radiation means that radiations possess particle-like and wave-like properties.Two developments played a major role in formulating the Bohr model of the atom. Let’s have a look at Bohr’s atomic model. Quantum mechanics serves as the foundation for the contemporary model of an atom. 
In 1913, Neil Bohr put forward an atomic model based on Planck’s quantum radiation theory. 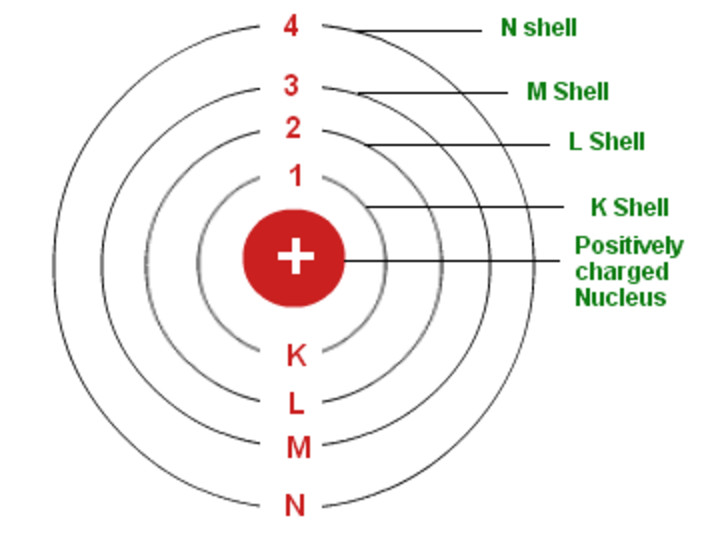
To remove the drawbacks of Rutherford’s nuclear atomic model and explain the origin of the production of the spectrum of Hydrogen and Hydrogen-like species (such as Li 2+, He +, etc.).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
